Biology
Nanosecond-resolution photothermal dynamic imaging via MHz digitization and match filtering
J. Yin, L. Lan, et al.
The study addresses the limitation of conventional photothermal microscopy that relies on lock-in amplification at the fundamental modulation frequency, which obscures rich transient thermal dynamics and higher-order harmonics. Photothermal heterodyne imaging (PHI) and mid-infrared photothermal (MIP) microscopy have enabled sensitive, label-free detection with submicron resolution and broad applications. However, lock-in demodulation loses temporal resolution and higher harmonics, limiting the ability to interpret complex signals from absorbers embedded in media with distinct decay lifetimes, and requiring repetitive measurements for time-gated approaches. The research question is whether lock-in-free detection with wideband digitization and matched filtering can directly capture nanosecond-resolved photothermal dynamics within single-pulse excitation, improve SNR and imaging speed, and separate overlapping signals (e.g., water background vs biomolecules) in heterogeneous biological samples.
Photothermal microscopy enables absorption sensing via thermal effects, achieving single nanoparticle and single-molecule sensitivity with lock-in-based heterodyne detection. MIP microscopy uses mid-IR pump and visible probe to achieve submicron resolution and molecular specificity, with advances including wide-field detection, optical phase detection, photoacoustic detection, and integration with Raman. Lock-in-based approaches can infer thermal diffusivity via phase but at the fundamental frequency lose higher harmonics and temporal detail, limiting dynamic range and quantitative interpretation in complex environments. Time-gated and optical gating methods resolve dynamics but require repetitive scans. Prior work used various mid-IR sources and detection schemes, but sensitivity and background separation remain challenges when signals with different decay constants overlap, such as water versus biomolecules in cells.
The authors model transient photothermal dynamics under pulsed mid-IR excitation using a heat transfer equation where temperature evolution depends on absorbed energy (Q_abs ~ I_IR(t)σ_abs) and dissipation (Q_diss ~ hS[T(t)−T_env]). For pulsed excitation shorter than the thermal relaxation time, heating and cooling follow exponential behavior with time constant τ = mC_p/(hS), analogous to an RC circuit. In the frequency domain, the photothermal waveform yields harmonics at multiples of the IR repetition rate; the frequency response amplitude is |H(f)| = 1/(1+(2πτf)^2), implying fast decays distribute energy into higher harmonics. Instrumentation: The PDI system uses a tunable QCL (900–1790 cm⁻1) as mid-IR pump (300–1000 ns pulses, 100 kHz–1 MHz repetition), focused with a reflective objective. A counter-propagating continuous-wave 532 nm probe is focused with a water-immersion objective. Forward and backward scattered probe light are collected on silicon photodiodes via beam splitter and dichroic. The photocurrent AC component is amplified with a wideband amplifier (10 Hz–100 MHz, 40 dB), low-pass filtered at 25 MHz, and digitized at 50 MS/s, 14-bit. An MCT detector monitors the IR pulse shape synchronously. The same amplified signal can feed a lock-in amplifier for comparison. Data processing: For each pixel dwell segment (as low as 50 µs), raw time traces containing multiple pulses are match-filtered in the frequency domain using a comb passband centered at harmonics of the IR repetition frequency (e.g., 100 kHz, 200 kHz, ... up to 1.6–25 MHz depending on task). After filtering, inverse FFT yields denoised temporal dynamics. Including multiple harmonics boosts SNR by coherently summing correlated signal harmonics while rejecting uncorrelated noise. X–Y–t stacks are reconstructed using synchronized stage position feedback. For spectra, photothermal intensity at t = 300 ns is normalized by effective absorbed energy (integrated IR pulse to 300 ns) and spot-size dependence. Thermal decay constants are obtained by nonlinear regression fitting of exponential decay in the post-pulse region; advanced estimators (MLE, maximum entropy) are noted as alternatives. Sample preparations include PMMA nanoparticles on CaF2, E. coli grown on 12C or 13C glucose, and U87 cells fixed and imaged in D2O-based buffer.
- PDI captures single-pulse photothermal signals with nanosecond resolution: single-pulse SNR > 43 (PMMA 500 nm at 1729 cm⁻1), improved to SNR 570 after matched filtering with 200 µs dwell.
- SNR improvement over lock-in: For 300 nm PMMA at 1729 cm⁻1, PDI achieved SNR 230 at 200 µs dwell versus 52 with lock-in demodulation at 100 kHz (≈4.3× improvement), close to theoretical 5.4× for the conditions used.
- Harmonic content: Transient signals contain significant high-frequency harmonics; matched filtering across harmonics improves sensitivity and preserves dynamics.
- Thermal dynamics quantification: For a 300 nm PMMA particle, decay constant τ ≈ 300 ns; derived heat transfer parameter hS ≈ 7.78×10⁻⁸ W/K, agreeing with FEM calculation (7.65×10⁻⁸ W/K). Across sizes, τ ≈ 280 ns (300 nm) and 495 ns (500 nm). Estimated peak temperature rise for 500 nm PMMA at 1729 cm⁻1 is ~7.6 K within <1 µs.
- Speed gains: Using τ to set repetition rate, imaging at 500 kHz (400 ns pulses) achieved 100 µs dwell with SNR ≈ 261 for 500 nm PMMA, ~30× faster than prior work with similar SNR.
- Single-bacterium metabolism imaging: For E. coli at Amide I (1658 cm⁻1), τ ≈ 280 ns enabling 1 MHz modulation and 50 µs dwell; single-cell SNR > 140; >350 cells imaged in 70 s (~600× faster than previous lock-in-based MIP). Isotope labeling shifts: Amide I from 1658 to 1615 cm⁻1 and Amide II from 1546 to 1533 cm⁻1 with 13C-glucose incorporation.
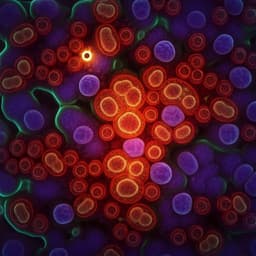
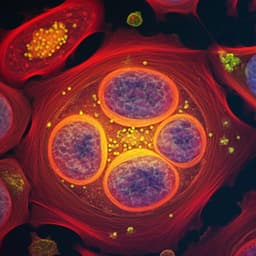
- Water background separation: In U87 cells, decay constants differentiate components—lipid droplets τ ~150–500 ns at 1750 cm⁻1, protein-rich cytoplasm/nucleolus τ ~2.5 µs at 1655 cm⁻1, water background τ >5 µs. Signals appearing at 1655 cm⁻1 in lipid regions are attributed to water-induced refractive index modulation rather than lipid absorption.
- High-order harmonics enhance lipid contrast: Lipid signals distribute energy into high harmonics whereas water background concentrates at the fundamental. At the 21st harmonic (2.1 MHz), small lipid droplets are clearly resolved with minimal background; lock-in at the 1st harmonic (100 kHz) shows poor lipid-to-background ratio.
- Spectral fidelity: PDI-derived spectra of PMMA match FTIR in the fingerprint region, preserving peak ratios.
The findings demonstrate that lock-in-free PDI with MHz digitization and matched filtering directly captures transient photothermal dynamics at nanosecond resolution, addressing the loss of temporal information inherent to lock-in detection. By coherently summing harmonic components where laser noise is lower, PDI substantially improves SNR for low duty-cycle photothermal signals and enables much faster imaging without sacrificing sensitivity. The approach reveals heterogeneous, chemically dependent thermal environments in cells: lipid droplets exhibit fast decays, while proteins and the aqueous medium show slower dynamics. Leveraging these distinct time constants, PDI separates weak biomolecular signals from overlapping water background and can target high-order harmonics to maximize contrast of fast-decaying absorbers. Compared to alternative demodulation schemes (multi-channel lock-in, boxcar), PDI captures the full harmonic content and provides accurate decay constants with broadly available hardware. The method extends the capabilities of MIP microscopy to characterize both chemical absorption and physical thermal properties, facilitating high-throughput single-cell metabolism studies and enabling detection of small organelles otherwise buried by background.
This work introduces a lock-in-free mid-IR photothermal dynamic imaging method that combines wideband detection, MHz-rate digitization, and matched filtering across harmonics to achieve nanosecond temporal resolution and submicron spatial resolution. PDI improves SNR by more than four-fold over conventional lock-in methods and accelerates imaging by up to two orders of magnitude. It quantifies thermal decay constants, estimates transient temperature rises, and separates biomolecular signals from water background in living cells by exploiting distinct decay dynamics. Demonstrations include accurate thermal characterization of PMMA nanoparticles, high-throughput single-bacterium metabolic imaging with isotopic shifts, and visualization of small lipid droplets via high-order harmonics. Future directions include applying PDI to study short-lived nonlinear photothermal phenomena (e.g., nanobubbles), higher-order thermal wave effects on heat capacity and conductivity, resistive heating in nanostructures, and fast cellular responses, as well as extending to other spectral regions and complex multi-lifetime decay analyses.
- Temporal resolution is ultimately limited by the photon detector (photodiode) response time (few nanoseconds).
- A single-exponential decay model may be insufficient for complex specimens; multi-exponential or distribution models may be required.
- Discrepancy between theoretical and experimental SNR gains arises from non-ideal IR pulse shapes.
- Practical imaging speed can be limited by the mechanical scanning stage (minimum dwell ~50 µs).
- Presence of water absorption necessitates buffer choices (e.g., D2O) and dynamic analysis to mitigate background.
- Optimal modulation frequency depends on the absorber’s decay; simultaneous optimal detection for mixed decay constants is challenging when using a single frequency in lock-in systems (mitigated in PDI by capturing full harmonics).
Related Publications
Explore these studies to deepen your understanding of the subject.